Techniques & Approaches
// We employ a diverse toolkit spanning molecular, cellular, circuit, and behavioral levels of analysis.
$ neurohab — Integrated Behavioral Arena
We developed the NeuroHab, an integrated behavioral arena that enables high-fidelity operant conditioning training and automated data collection in a single unified system. Operant elements such as food and water delivery as reward, conditioned stimulus, and event recording are tied together programmatically with easy-to-install open-source code to facilitate throughput and reproducibility.
// Figure 14. NeuroHab with all Mods installed. Home (left) Core (right).
$ timing_precision
All behavioral events — including food delivery, water delivery, and conditioned stimuli — are processed by internal microcontrollers and logged with <1 ms latency (typical sensor-to-log range: 56–728 μs under normal operating conditions). This precise timing is critical for integrating our system with two-photon imaging and electrophysiology equipment, enabling us to align behavior with brain activity in real time. These timestamps allow us to associate behavior with neural activity, such as calcium events and neuronal spikes, with high precision.
$ hardware_modules
The NeuroHab uses solenoid-actuated, capacitive-sensing Lickports to control lick detection and water delivery, enabling an untethered mouse to drink from an automated port similarly to a standard home-cage water bottle. For food delivery, we utilize the Kravitz Lab FED3, which detects nose pokes to automate pellet dispensing. Conditioned stimuli are delivered by dedicated modules, each containing a buzzer and an RGB LED.
$ core_architecture
All stimulus delivery is automated and recorded by the central control system, known as the Core. The NeuroHab Core coordinates all modules and records their outputs. It uses TTL pulses for communication between its two microcontrollers to orchestrate the behavioral tasks and log all event timestamps in chronological order.
$ surgery_and_viral_injections
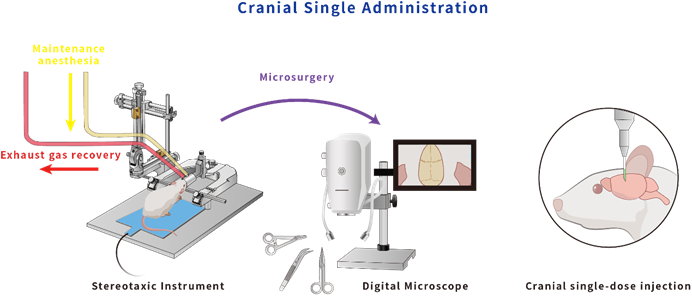
Employing rigorous surgical procedures and viral vector injections, we modulate neural circuits and gene expression patterns to elucidate their roles in behavior and disease progression.
// Stereotaxic surgery and viral injection workflow
$ viral_workflow
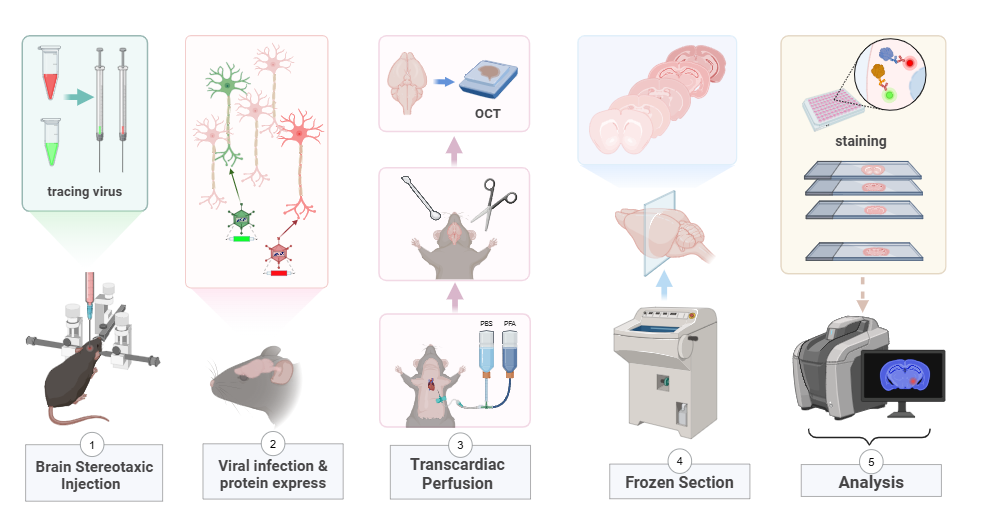
Using advanced genetic techniques, we create mouse models targeting specific cell types and marker genes, enabling precise manipulation and observation of cellular processes.
// Brain viral expressing workflow (confocal)
$ electrophysiology
Single-unit extracellular recordings, simultaneous multiple patch-clamp, local field potential (LFP) recordings, and in vivo whole-cell recordings.
// SliceScope Pro electrophysiology rig
$ patch_clamp
Simultaneous multiple patch-clamp recording system for decoding complex neural circuits with optogenetic assistance.
// Patch Pro 1000 electrophysiology rig
$ two_photon_imaging
Multi-photon imaging and fiber photometry to capture real-time calcium activities in vivo, enabling high-resolution imaging of neural dynamics.
// Prairie Ultima IV two-photon in vivo rig
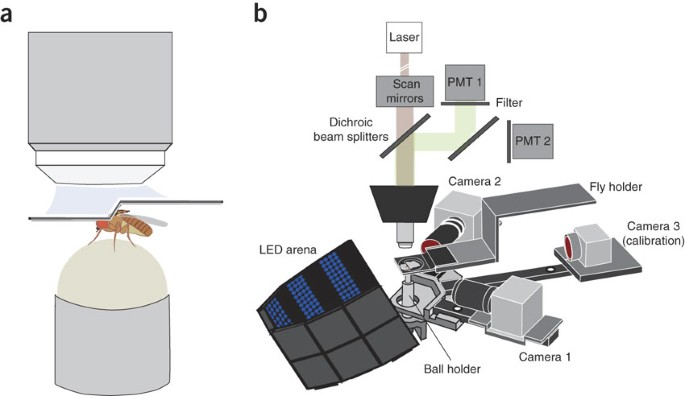
$ in_vivo_imaging
In vivo imaging setup for capturing neural activity during awake behaving experiments with head-fixed animals.
// Two-photon calcium imaging setup diagram
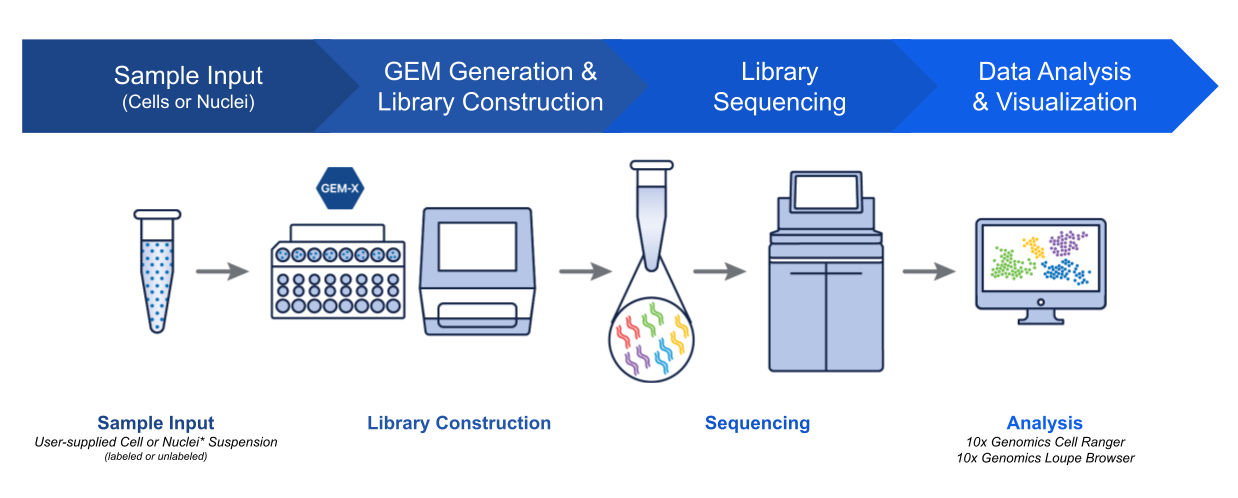
$ single_cell_rnaseq
Leveraging platforms like 10x Genomics, we perform single-cell RNA sequencing to dissect cellular heterogeneity and gene expression profiles within neural populations.
// 10x Genomics GEM-X scRNA-seq workflow
$ scrnaseq_pipeline
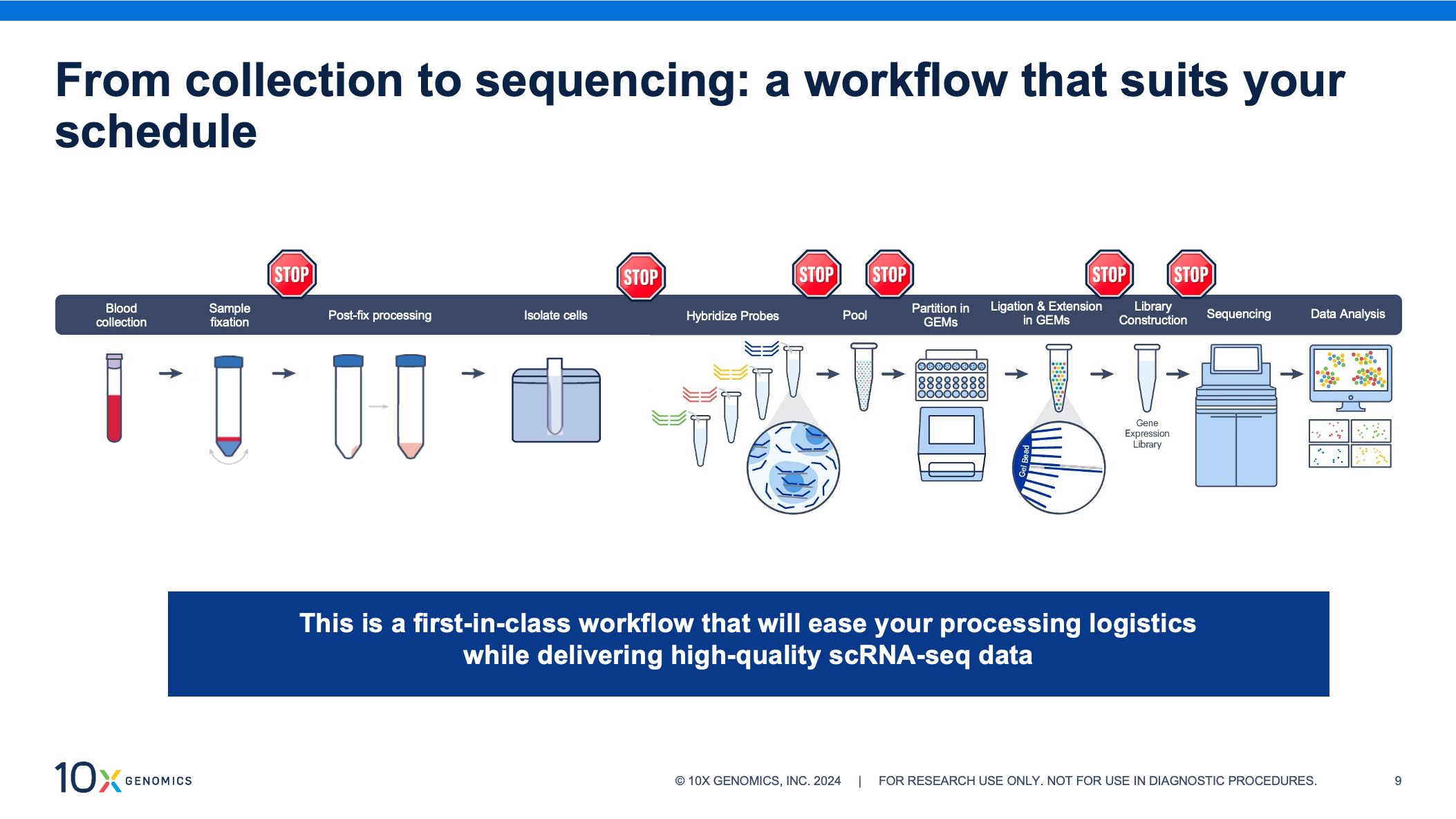
Complete pipeline from sample collection to sequencing and data analysis for high-quality single-cell profiling.
// From collection to sequencing workflow
$ behavioral_assays
Awake behaving mouse paradigms, persistent licking tasks, wheel running, sensory discrimination, and seizure monitoring during sleep.
$ circuit_tracing
AAVretro viral tracing, rabies-based monosynaptic tracing, anterograde and retrograde labeling of long-range projection neurons.
$ computational_modeling
Network simulations of E-I balance, computational models of persistent activity, and analysis of spike train dynamics and synchrony.
$ optogenetics
Channelrhodopsin-2 (ChR2) activation, opto-tagging of projection neurons, subcellular Laserspritzer stimulation, and near-infrared optogenetic tools.
$ histology_imaging
Histological techniques, immunohistochemistry, and confocal imaging to visualize cellular structures and molecular markers in tissue samples.
$ population_decoding
Decoding neural signals to infer cognitive states and behavioral patterns from population-level activity.